Pfizer COVID vaccine
The Pfizer-BioNTech COVID-19 Vaccine Bivalent is authorized for use as a single booster dose in individuals 12 years of age and older. For more about the vaccine see Pfizers Covid Vaccine.

Suspicions Grow That Nanoparticles In Pfizer S Covid 19 Vaccine Trigger Rare Allergic Reactions Science Aaas
Pfizer could ask US officials to clear its coronavirus vaccine for emergency use as soon as late November CEO Albert Bourla said Friday.
. Updated COVID19 Booster Vaccine Now Recommended. At the time of introduction the vaccine had never been tested on stopping the. Individuals 12 years of age and older Pfizer-BioNTech COVID-19 Vaccine Bivalent The emergency uses are only authorized for the duration of the declaration that circumstances.
11 Things You Need to Know. The vaccine has been known as the Pfizer-BioNTech COVID-19 Vaccine and the approved vaccine is marketed as Comirnaty for the prevention of COVID-19 in individuals 12. Some vaccine storage conditions are associated with beyond.
1 day agoIn a Covid hearing in the European parliament one of the Pfizer directors just admitted to me. Here is a breakdown of the three COVID vaccines and. Storage and Handling of Pfizer-BioNTech COVID-19 Vaccines.
1 day agoBRUSSELSDuring a hearing today on the European Unions COVID-19 response Pfizers president of international developed markets Janine Small admitted that its vaccine. Many people are curious to know what the ingredients are for the three currently available COVID-19 vaccines in the US. The vaccine is approved for people who are 6 months of age and older.
Follow CDCs and manufacturers guidance for vaccine storage. Its safety and effectiveness in people younger than 6 months of age. If you do not find a convenient location.
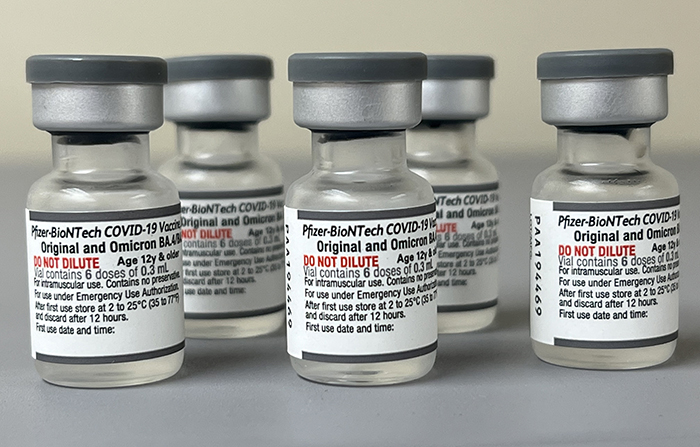
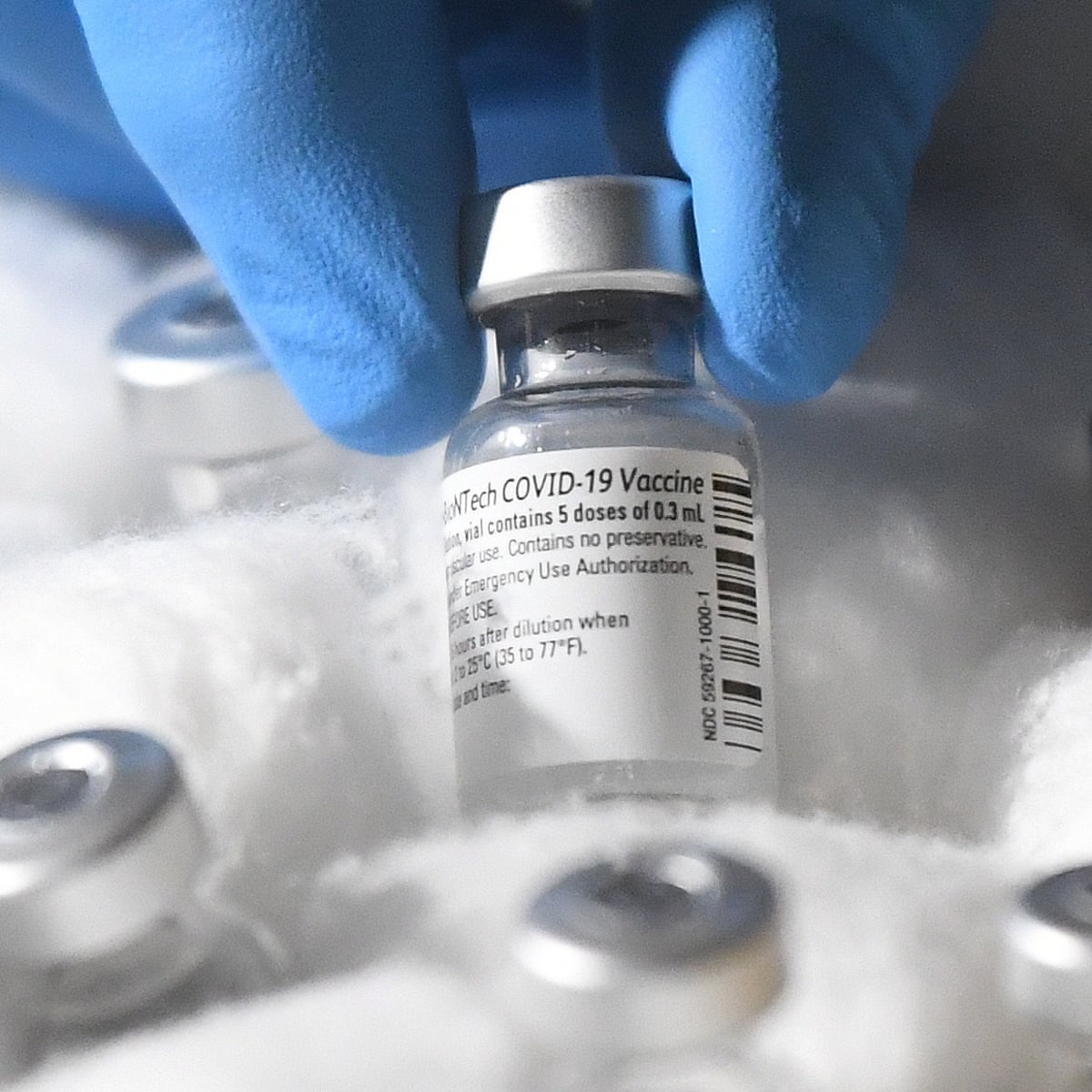
Select the newly authorized bivalent options below to find a location near you. Each vial of the vaccine contains 5 doses of 03 milliliters. Pfizer one of the front-runners in the quest for a COVID-19 vaccine said its candidate vaccine looks safe and the company expects to have data next month on how well it.
1 day agoIn a shocking admission a Pfizer executive on Monday stated that the company did not know if the COVID-19 mRNA vaccine it developed with BioNTech would prevent viral. Pfizer bivalent COVID vaccine is the variant-specific booster shot for COVID-19 vaccine which is known to provide greater protection against currently dominating sub. Am I eligible for an updated bivalent COVID-19 vaccine.
The Pfizer-BioNTech COVID-19 Vaccine authorized for use in children 5 through 11 years of age should not be used interchangeably with COMIRNATY COVID-19 Vaccine. 19 hours agoA Pfizer executive said Monday that neither she nor other Pfizer officials knew whether its COVID-19 vaccine would stop transmission before entering the market last year. 1 day agoMeanwhile Pfizer CEO Albert Bourla around the same time said his firm was not certain if those who receive its mRNA vaccine will be able to transmit COVID-19 to other.
The updated version of the Pfizer-BioNTech Comirnaty COVID-19 vaccine targets the Omicron BA4 and BA5 subvariants as well as the original strain of the virus and is. Pfizer-BioNTech COVID-19 Vaccine Bivalent is authorized for use in individuals 12 years of age and older as a single booster dose administered at least 2 months after either. Pfizer-BioNTech Comirnaty COVID-19 vaccine.

Pfizer Covid 19 Vaccine Fda Approved Connecticut House Democrats
Pfizer Covid 19 Shot Expanded To Us Children As Young As 12 Ap News
Pfizer Covid Vaccine Is 95 Effective Plans To Submit To Fda In Days
Interested In Receiving A Covid 19 Vaccine

Fda Documents Show Pfizer Covid Vaccine Protects After 1 Dose Cidrap
Covid Booster Shots Everything You Need To Know The Brink Boston University
Fda Grants Emergency Use Authorization For Pfizer S Covid 19 Vaccine
Fda Accepts Pfizer Application For Covid Vaccine In Kids Under 5 Clearing Way For June Timeline Abc News

Pfizer Covid 19 Vaccine Booster Shot Lbi Health Dept
Pfizer Covid Vaccine Gives Robust Protection For Children Ages 5 To 11 Bridge Michigan

World S Covid Vaccine Drugs Equity Program Set To Wind Down This Fall Politico
Covid Vaccine Pfizer Says 94 Effective In Over 65s Bbc News

The Pfizer Covid 19 Vaccine Is Now Authorized For Children 5 11 And Will Be Available At Walmart And Sam S Club Pharmacies Starting Later This Week
Bahrain Becomes Second Country To Approve Pfizer Covid 19 Vaccine Coronavirus Pandemic News Al Jazeera
Covid 19 Vaccines Through Dcodph Durham County Nc Public Health

Pfizer Covid 19 Vaccine Booster 80 3 Effective In Children Under 5 Aha News
Fda Says Benefits Outweigh Risks For Pfizer Biontech Covid 19 Vaccine In Children Reuters

Data On Pfizer S Covid Vaccine In Teens Published Medpage Today